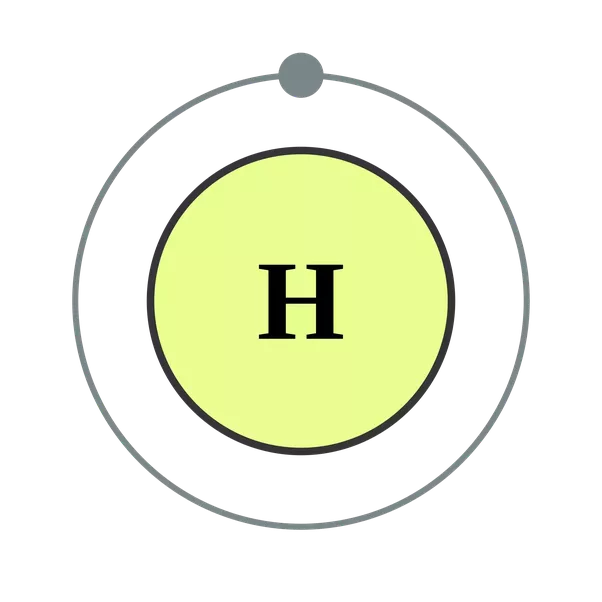
Atoms are way too small to see with the naked eye (and even most microscopes). It features a 1-hour lecture video, and also presents the prerequisites, learning objectives, reading assignment, lecture slides, homework with solutions, and resources for further study. In effect, an atom can “store” energy by using it to promote an electron to a state with a higher energy and release it when the electron returns to a lower state. An early model of the atom was developed in 1913 by Danish scientist Niels Bohr (18851962). In atomic physics, the Bohr model if the atom (also known as the Rutherford-Bohr model) is modern model of the hydrogen atom introduced by Danish physicist Niels Bohr working with Ernest Rutherford at the University of Manchester in 1913. The Bohr model (article) Khan Academy > Science > High school chemistry > Atoms, elements, and the periodic table > The Bohr model and atomic spectra The Bohr model Google Classroom Learn how Bohr models are used to represent atoms. This page contains materials for the session on the atomic models of Rutherford and Bohr.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
